If you or a loved one are currently taking Concerta for ADD/ADHD treatment, you need to know about this change from pharmaceutical manufacturers. Terri West, PA-C, Associate Medical Director with the Being Well Center, shares everything you need to know to discuss further with your doctor to protect the reliable treatment you know and trust.
The FDA recently approved 3 new generics for Concerta from the following pharmaceutical manufacturers:
- Trigen Laboratory
- Mylan Laboratory
- Impax Laboratory
Effective January 1, 2018, many big chain pharmacies have made the decision to carry only one of the 3 new generic Concerta options. After these pharmacies deplete their current supply of Actavis, they will no longer carry the “authorized” generic Concerta with the unique OROS delivery system. We recommend the OROS delivery system because many years of clinical experience have taught us it is the most effective methylphenidate treatment for ADD.
Pharmacists and medical professionals who are unaware of the unique properties of the OROS system used in brand Concerta and the “authorized” or “true” generic form Actavis may assure patients that these new generics are the same as Concerta and may substitute your prescription with any of the three new products despite our directions that state, “OROS Delivery System Only, Actavis generic only” on our written or electronic prescriptions.
Health insurance plans or their associated Prescription Benefits Managers (PBMs) determine which brand or generic medications are on your formulary and whether they are “preferred” or “non-preferred.” Prescriptions must be written as “brand” if brand is preferred on your formulary. When plans list “generics” as “preferred” on your formulary, the prescription may be filled by a pharmacist with any FDA AB rated (meaning it has the same active ingredient as the brand) approved generic medication unless the prescribing provider specifies OROS Delivery System Only by Actavis. It is the OROS delivery system that sets Concerta apart from other stimulant medications.
Insurance plans do not stipulate which generic your pharmacist gives you or if the generic has the OROS delivery system. To be certain that you receive the “authentic generic,” the prescription must specify “OROS Delivery System Only” by Actavis and your pharmacy must carry the generic by Actavis, or an osmotic delivery system that is equivalent to brand Concerta.
At this point, we have no way of knowing if any of the 3 new generics have the OROS delivery system equivalent to the brand Concerta or the Actavis generic. Therefore, we do not know if the efficacy and duration of the new generic products are equivalent to the brand or the “authorized” generic by Actavis. Some patients given the new generics may find that they work well, while others may experience decreased efficacy or have unpleasant side effects.
The bottom line is that the “authorized” generic by Actavis may no longer be an option at your pharmacy or through mail-order services if the pharmacy does not carry the Actavis generic. The Actavis Laboratory continues to manufacture their “authorized” OROS delivery system generic (methylphenidate Hcl ER) for Concerta and as of January 10th, 2018, they have assured me that they will continue producing and shipping it to major distributors throughout the US.
Up until the end of 2017, most large drugstore chains carried the Actavis “authorized” generic Concerta.
At that time, some of the chains had corporate policy changes mandating their pharmacies to exclusively carry only one of the 3 newer generics. We know that the major pharmacies in the Tri-State area exclusively carry the following new generic extended release methylphenidate products:
- CVS: Trigen generic
- CVS Caremark mail-order: Trigen generic
- Rite Aid: Mylan generic
- (no local pharmacies): Impax generic
The local Pittsburgh pharmacies that still carry the “authorized” generic OROS by Actavis include:
- Walgreens has the “true” generic OROS by Actavis
- Walmart has the “true” generic OROS by Actavis
- Medicine Shoppe, Ingomar, PA has the “true” generic OROS by Actavis
- Independent pharmacies may or may not carry the Actavis generic
The Medicine Shoppe in Ingomar, owned and operated by Dave Wessel, R Ph., continues to carry the Actavis generic Concerta. However, restrictions from pharmacy distributors may soon limit The Medicine Shoppe and other independent pharmacies who carry the Actavis product from being able to keep an adequate supply in stock for patients when their prescriptions are due.
If your pharmacist has given you one of the new generic substitutions for brand Concerta or Actavis OROS generic, please alert our office staff ASAP so that we can document the name of the new manufacturer (Mylan, Impax,Trigen) and monitor the efficacy of your treatment regimen.
If you experience any side effects or problems on the new generic or feel that the effectiveness or duration is not the same as your prior medication, please contact your insurance plan first and ask if they will cover brand Concerta because you are not tolerating the new generic substitution medication. Then call The Being Well Center with the information so we can discuss what options may be available to you.
For all new patients doing medication trials on Concerta, we prefer that you do NOT accept any of the new generic medications until you are on a therapeutic and stable treatment plan.
The Being Well Center will continue to advocate for our patients with the goal being an individualized therapeutic treatment plan with the proper medication for all day coverage along with the necessary supports from counseling to each individual’s unique needs.
We will update patients/families as new information becomes available. Please check our website www.thebeingwellcenter.com and our Facebook page for more updates regarding the OROS delivery system and the new generic substitutions for Concerta. If you have specific questions regarding this information, please contact the BWC and ask to speak with Terri West, PA-C.
What is the OROS Delivery System and why is it so important?
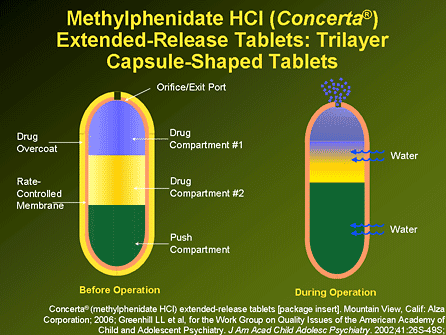
Concerta has a unique delivery system (OROS) that uses osmotic pressure to deliver methylphenidate through the small drill hole in the tablet at a controlled and predictable rate. The outer colored coating of the hard tablet dissolves and provides the initial dose of medication within the first hour. Water in the gastrointestinal tract permeates through the outer hard tablet shell, causing the “push” component to expand in the end of the tablet, pushing the methylphenidate out through the tiny drill hole at the opposite end of the tablet (see diagram) at a controlled rate over a 12 hour period. The hard outer shell does not dissolve; the tablet remains intact as it passes through the gastrointestinal tract and is eliminated in the stool. The brand Concerta and Actavis generic tablets can also be seen on plain abdominal x-ray films.
History of Concerta
Brand Concerta FDA Approved in 2000
In 2013, the manufacturer of brand Concerta made an agreement with Watson Labs to manufacture their patented Concerta OROS drug in the Watson Lab and sell it as an “authorized” or “true” generic drug for Concerta at a lower cost. The drug was exactly the same as the brand and was available in pharmacies as a generic by Watson Labs. Watson was later bought out by Actavis Labs who continue to manufacture the “authorized” generic for distribution.
Concerta (OROS Delivery System) = “authorized” generic by Actavis
Shortly after the “authorized” OROS generic came out, 2 other generics were released by Mallinckrodt Laboratories and Kremer Laboratories (Kremer later changed names under Kudco Labs or Lannett Labs). Both generic methylphenidate hcl ER were approved by the FDA as “AB” rated generics equivalent for Concerta. Their product insert submitted to the FDA was essentially the same description as brand Concerta OROS delivery system, therefore, it was confusing to pharmacists when we insisted that the generic was not equivalent to the brand or Actavis generic.
The tablet shape was oblong and similar to Concerta and the color for each different mg dose corresponded to brand Concerta. Only Individuals who were very familiar with the OROS system might have noticed that the new generics were missing a critical feature: the tiny drill hole in the tablet where the methylphenidate is released. Instead, when this generic was exposed to water the outer coating/shell partially dissolved, exposing the methylphenidate inside to dissolve in the GI tract. It did not have an OROS system and no hard shell was eliminated in the stool. The active ingredient, methylphenidate was the same as in Concerta but the method of delivery and the bioavailability of the methylphenidate was clearly different.
The difference was not apparent at first as some patients had prescriptions that were filled with half of the Actavis generic and half of the Mallinkrodt or Kremers generic.Patients stable on the same Concerta dose for years were presenting to their physicians with multiple side effects that resolved when placed back on the brand or Actavis generic for Concerta. Many physicians and patients began reporting problems with the 2 new generic medications to the FDA via phone calls or through the online FDA website.
The reports led to an FDA investigation regarding the bioequivalence of the new generics compared to brand Concerta in November of 2014. Generic drugs have the same active ingredient as brand medications. However, the method in which the active ingredient is released into the bloodstream (ie: dissolving tablets, extended release beads, injection, OROS system), determines the speed at which the medication is released and may affect the blood level of the active drug or alter the duration of a medication. This is especially important in stimulant medications, when even a small change in the dose or timing of dose is critical.
If the availability and duration of effect of the drug are not the same, the generic is downgraded to a BX rating. BX rated drugs are not to be substituted for brand name drugs. The FDA states that pharmacists are not permitted to substitute Mallinckrodt or Kremers generics for Concerta at the pharmacy unless the prescribing physician/provider specifically requests or agrees to use the “BX” rated generic.
Unfortunately, the first 2 generics are still available at some pharmacies and prescriptions may be filled with an unacceptable substitute for Concerta, when in fact it is not.
These generics are not FDA approved for Concerta:
- Kremer/Kudco/Lannett for Concerta
- Mallinckrodt generic for Concerta
Do not accept this generic with an “M” stamped on the tablet.
Always check your prescription bottle before leaving the pharmacy!
Mallinckrodt generic

The New Generics 2017- 2018: Know the Options
All three of the new generics claim to be the same as brand Concerta, with an OROS delivery system, and they are ‘AB’ rated and approved by the FDA. This does NOT ensure that the 3 new generics have the same unique OROS delivery system or that they are therapeutically equivalent. The 3 manufacturers claim they have an ‘osmotic’ system, which is not to be confused with OROS Delivery System. There is no way to know if their product delivers the medication as precisely and consistently as the OROS delivery system in brand Concerta and the “true” generic by Actavis.
Mylan generic* Trigen generic
 *Note that 8 of the 18 pills do not have drill holes
*Note that 8 of the 18 pills do not have drill holes
This is a picture of the Mylan generic from a patient who experienced an adverse reaction.
MedWatch Reporting of Adverse Events
Reporting adverse events (problems related to a medication) to the FDA is important for both patient/parent and healthcare providers. It is the most effective way to demonstrate a variability or problems with a new or generic medication. The reports must be accurate and all parts of the form must be completed in order for the FDA to utilize the information in their data bank and to determine whether the adverse event meets the criteria for further investigation. In regard to Concerta generics, reporting will also help to alert and educate both consumers and medical professionals who are unaware of the importance of the OROS delivery system in Concerta and the “authorized” Actavis generic.
PLEASE take the time to report any problems with the generic medication.
Please go to the FDA website and search for MedWatch to locate the form: “Consumer Reporting of Adverse Events 3500”
Complete the online report and submit it to the FDA or download the form to complete and mail in. For our Being Well Center patients: Please bring the form to the BWC if you would like help in completing the report. Our staff can quickly help you to complete the paperwork, make a copy for your records, and mail it to the FDA.
The form is easy to complete. Aside from a description of what you experienced while taking the medication you will also need the following information to complete the MedWatch form:
- Name of medication as it appears on the bottle
- Name of company or manufacturer of drug
- Date on bottle
- Date medication filled
- Quantity of pills on prescription label
- Mg dose of tablet/capsule
- Lot number (call pharmacist to inquire about the lot # that your prescription was filled)

ABOUT THE AUTHOR: Terri West is a NCCPA board certified Physician Assistant who received her training at Gannon University. She has over 20 years of experience in working with patients across the lifespan in a variety of settings including pediatrics, family practice, gynecology, surgery, and a breast cancer specialty practice, where she gained a keen insight into women’s health issues.























